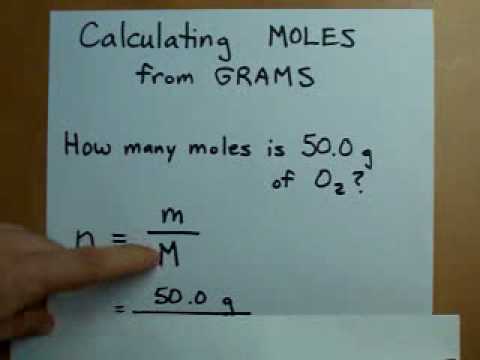
1 mole is equal to 1 moles In, or 114.818 grams. You can view more details on each measurement unit: molecular weight of In or grams The SI base unit for amount of substance is the mole. We assume you are converting between moles In and gram. In chemistry, the molar mass of a chemical compound is defined as the mass of 1 mole (or 6.02214×10 23 particles) of the substance, expressed in grams. 1 mole is equal to 1 moles O2, or 31.9988 grams. You can view more details on each measurement unit: molecular weight of O2 or grams The SI base unit for amount of substance is the mole. We assume you are converting between moles O2 and gram.
#Grams to moles free#
You can also verify the results by putting the values in free grams to molecules calculator. Carrying out grams to moles conversion: n m M.

Enter the Molar Mass in the second field. You may want to calculate this value as a double-check of your mastery of calculating molar. 2) The molar mass is 34.0146 grams/mole, which is calculated using the formula and the atomic weights on a periodic table. Solution: 1) 17.0 grams are given in the text of the problem. You need to follow the simple instructions to find all grams to moles given below. Example 2: Calculate how many moles are in 17.0 grams of H 2 O 2. Step 2: Divide the number of grams of the compound by its molecular mass. Use the periodic table to check the atomic mass, this is the number of grams per mole. Converting grams to moles involves 2 steps: Step 1: Find the molecular mass of the compound. Molar Mass of 5 Litres of water 5 18.015. Using grams to moles calculator is the easiest ways to calculate moles to grams or grams to moles. There are three steps to converting grams of a substance to moles. You can view more details on each measurement unit: molecular weight of Oxygen or mol The molecular formula for Oxygen is O.įurthermore, how many grams is 1.667 moles of oxygen? 1 mole is equal to 1 moles Oxygen, or 15.9994 grams. Now we have: Molar Mass of 1 Litre of water 18.015. We assume you are converting between grams Oxygen and mole. 1 grams O2 is equal to 0.031251171918947 mole.Ĭorrespondingly, how many moles are in 28.0 grams of oxygen? You can view more details on each measurement unit: molecular weight of O2 or mol The SI base unit for amount of substance is the mole. We assume you are converting between grams O2 and mole.
